J Pharm Pharmaceut Sci (www.ualberta.ca/~csps) 4(2):176-184, 2001
An In Vitro Evaluation of Human Cytochrome P450 3A4 and
P-glycoprotein Inhibition by GarlicBrian C. Foster1,
Office of Science, Therapeutic Products Directorate, Health Canada, Ottawa, Ontario, CanadaMeredith S. Foster, Susan Vandenhoek, Anthony Krantis
Centre for Research in Biopharmaceuticals, University of Ottawa, Ottawa, Ontario, CanadaJason W. Budzinski, John T. Arnason
Ottawa-Carleton Institute of Biology, University of Ottawa, Ottawa, Ontario, CanadaKeith D. Gallicano
Axelson Biopharma Research, Inc., Vancouver, British Columbia, CanadaShurjeel Choudri
St. Boniface General Hospital, Winnipeg, Manitoba, CanadaReceived January 15, 2001, Revised June 29, 2001, Accepted June 30, 2001
PDF version
Abstract
Purpose. Garlic has been used as a flavouring agent, traditional medicine, and functional food to improve physical or mental well-being. Garlic and garlic products generally have been regarded as safe but a number of conflicting reports in the literature and confounding factors make it difficult to unequivocally establish the clinical efficacy and safety of these products either alone or in the presence of therapeutic products. A preliminary study was undertaken with fresh garlic and garlic products using the major cDNA-expressed human cytochrome P-450 isozymes associated with the metabolism of HIV/AIDS drugs, and purified P-glycoprotein (P-gp) cell membranes to ascertain the risk potential for generating interactions with therapeutic products.
Methods. A broad screening was undertaken with 10 garlic products (aged, odourless, oil, freeze-dried) and 3 varieties of fresh garlic bulbs (common, Elephant and Chinese), all purchased from local outlets, to examine their potential to affect human cytochrome P-450 2C9*1, 2C9*2, 2C19, 2D6, 3A4, 3A5 and 3A7 mediated-metabolism of marker substrates using an in vitro fluorometric microtiter plate assay. Four garlic products were screened for their potential to interact with P-gp using an in vitro colourmetric ATPase assay.
Results. Extracts of fresh garlic, different brands and lots of odourless garlic and representative samples of garlic oil, freeze dried garlic, and aged garlic exhibited an inhibitory effect on cytochrome P450 2C9*1, 2C19, 3A4, 3A5 and 3A7 mediated metabolism of a marker substrate. The activity of 2D6 mediated-metabolism was generally unaffected by garlic. Extracts of the fresh garlic stimulated CYP2C9*2 metabolism of the marker substrate. With the extracts tested, garlic had very low to moderate P-gp interaction as compared with the positive control verapamil.
Conclusions. Our in vitro findings demonstrate that garlic components can affect cytochrome P-450 2C, 2D and 3A mediated-metabolism of the isoforms studied. The safety and efficacy of conventional therapeutic products metabolized by the affected isozymes, particularly those with a narrow therapeutic index, taken concomitantly with garlic needs to be examined further under clinical settings.
Introduction
In the last decade in North America, a large proportion of the population has adopted botanical medicines to help manage or prevent the onset of chronic disease, improve cognitive function and well-being, and increase longevity (1). Herbal medicines reportedly have significant pharmacological activity and are capable of producing both potent therapeutic effects and adverse reactions (2). Literature and anecdotal reports suggest that concomitant oral administration of some natural products and pharmaceuticals may affect human drug metabolism and significantly increase the risk for serious clinical adverse reactions (3-12).
The main concerns lie primarily with the possibility of unknown natural product-drug interactions (13), possible over use because herbal products are self administered, and product variation due to the lack of standardization within the industry (14,15). For example, two HIV+ patients taking ritonavir experienced serious dose-dependent adverse events including nausea, vomiting, and diarrhea after taking either fresh garlic or odourless soft liquid-filled garlic supplements (8). Fresh garlic and an odourless soft liquid filled garlic supplement produced a dose-related inhibitory response on the formation of 6ß-hydroxytesterone from testosterone using microsomes containing human cytochrome P450 3A4 (16). A subsequent small clinical trial examined the effect of this odourless garlic supplement on single dose ritonavir pharmacokinetics in 10 healthy volunteers (17). There was a trend towards a lower area-under-the-concentration-time-curve (AUC) for ritonavir and a higher clearance in the presence of the garlic supplement, but the AUC decrease was not statistically significant (p = 0.094). These reports are of concern since a recent study conducted in presurgical patients showed that garlic was among the top 5 products taken in the preoperative period (18).
Fresh garlic ( Allium sativum ) and the 4 main categories of commercial preparations: oils of steam-distilled garlic, garlic macerated in vegetable oils, garlic powder, and gelatinous suspensions (14) have a variety of reported therapeutic properties including antimicrobial activity (bacterial, fungal and viral), antiplatelet aggregation, antihypertensive, and cholesterol lowering, which contribute to its increased usage as an alternative medicine (19). Allicin (diallyl thiosulfinate) is believed to be one of the major bioactive components of garlic (14). Allicin is formed by alliinase cleavage of the naturally occurring alliin upon crushing or mincing of garlic, and is the progenitor of a number of other products (14). Both alliin (approximately 1% in fresh garlic) and alliinase are stable when dry but allicin is heat labile and in the presence of organic solvents decomposes to form a variety of other organic sulfur products.
Lawson et al . (14) conducted an extensive phytochemical analysis of organic sulfur compounds of representative fresh and commercially available garlic products and found a wide variation in composition and chemical profile of sulfur compounds. Garlic powders suspended in a gel did not contain detectable amounts of non-ionic sulfur compounds. Thiosulfinates were only recovered from garlic cloves and powders. Vinyldithiins and ajoenes were only detected in garlic macerated in vegetable oil. Diallyl, methyl allyl, and dimethyl sulfides were exclusively found in oil of steam-distilled garlic. Typical steam-distilled garlic oil products contained similar amounts of total sulfur compounds as the total thiosulfinates released from freshly homogenized cloves; however, oil-macerated products contained about 20% whereas the garlic powders varied from 0-100%. Garlic is aged to reduce the content of sulfur compounds such as alliin and the odour commonly associated with garlic. Gel and aged garlic in aqueous ethanol products did not have detectable levels of these non-ionic sulfur compounds. Analysis of thiosulfinates from various Allium sp. revealed a 3-fold order of magnitude variation among species (15). Common garlic ( A. sativum ) and wild garlic had the highest levels, while elephant garlic ( A. ampeloprasum ) and Chinese chives have intermediary levels. Environmental conditions were also found to affect the total thiosulfinate levels.
Several garlic non-ionic sulfur compounds and their derivatives have been reported to affect drug disposition enzymes. Garlic oil suppressed inducible rat 2E1 expression more significantly than constitutive expression (20). The garlic oil tested was also found to induced glutathione S-transferase and microsomal epoxide hydrolase activity (20). When rats were treated with various garlic derived alkyl sulfides, the methyl derivatives had little or no effect (21). Compounds with two propyl or allyl groups such as diallyl sulfide and diallyl sulfone induced the amount of rat of 2B1,2 but inhibited 2E1 (21). The diallyl sulfone was reported to effect a suicide-substrate inhibition of rat 2E1 (22). Diallyl disulfide was preferentially metabolized by human liver microsomes to allicin by 2E1 with minor participation of other CYP isoforms and flavin-containing monooxygenases (23). In addition, aqueous extracts of garlic were shown to inhibit the pig liver microsomal activity of NADPH-cytochrome P450 reductase and NADH-cytochrome b5 reductase activity (24).
The objective of this study was to establish the potential for fresh garlic and commercially available garlic products (Table 1.) which may affect cDNA-expressed human cytochrome P-450 2C9*1, 2C9*2, 2C19, 2D6, 3A4, 3A5 and 3A7-mediated metabolism of marker substrates to ascertain the risk potential of these products to generate possible interactions with therapeutic products. As this was a preliminary screen, no attempt was made to equalize garlic marker concentrations of these non-ionic sulfur compounds.
Table 1. Characteristics of the commercial garlic products studied.
#
Amount of Garlic
(equiv. fresh garlic)Recommended Dose
(total daily)Additional Ingredients
1
5 mg powder odourless
(500 mg)2 cap / d
(1 g)soyabean oil, wax (yellow wax, hydrogenated veg. oil and veg. shortening), lecithin, Cap - gelatine, glycerine, water
2
5 mg powder odourless
(500 mg)3 cap / d
(1.5 g)soyabean oil, USP gelatine, veg. oil, water, lecithin
3
5 mg powder odourless
(500 mg)4 cap / d
(2 g)soya oil, lecithin, wax mixturecap - gelatine, glycerine, water
4
5 mg powder odourless
(500 mg)4 cap / d
(2 g)soya oil, lecithin, wax mixturecap - gelatine, glycerine, water
5
5 mg powder odourless
(500 mg)4 cap / d
(2 g)soya oil, lecithin, wax mixturecap - gelatine, glycerine, water
6
15 mg powder odourless
(1500 mg)3 cap / d
(4.5 g)soyabean oil, wax mixture, lecithin cap - gelatine, glycerine, water
7
3 mg oil
(1500 mg)1-2 cap / d
(1.5-3 g)veg. oil, gelatine, glycerine
8
920 mg oil
(460 mg)3 cap / d
(1.38 g)soyabean oil, gelatine, glycerine USP, purified water USP
9
400 mg freeze dried(4120 mg - TAP)
3 tab / d
(1.2 g)dicalcium phosphate, magnesium stearate
10
350 mg aged extract
1 cap / d
(350 mg)microcrystalline cellulose, magnesium stearate
Materials and Methods
Substrates and Reference Compounds
The fresh garlic and the garlic products (capsules and tablets) were obtained from local commercial outlets. These were assigned accession numbers and vouchers stored in the herbarium, University of Ottawa. The marker substrates: 7-Benzyloxyresorufin (7BR) and 7-ethoxy-3-cyanocoumarin (7EC) were obtained from Molecular Probes, Eugene, OR; 7-methoxy-4-trifluoromethylcoumarin (7-MFC) was obtained from Fluka Chemicals (Sigma), and 3-[2-(N,N-diethyl-N-methylamino)ethyl]-7-methoxy-4-methylcoumarin (AMMC) was obtained from GENTEST Corp. All other chemicals and solvents were of analytical grade.
Fresh garlic was peeled and weighed. A 1 g/ml aqueous solution was blended in a Waring blender until a uniform consistency was achieved and then polytroned for 1 min. Homogenates were placed in either 15 ml glass screw top vials or 1.5 ml microfuge tubes and placed in a -20°C freezer. Working solutions (25 mg/ml) were made from these stock solutions.
Garlic tablets were crushed using a mortar and pestle. The powders were made to a 25 mg/ml solution. The solution was vortexed on high for 1 min and then centrifuged for 18 min at 13,000 rpm. The supernatant was carefully removed for testing. Liquid capsules were cut open and the contents emptied into a 1.5 ml microfuge tube. The contents were suspended in 3 volumes/weight with 55% ethanol and vortexed for 1 min. The mixture was centrifuged for 18 min at 13,000 rpm. Four distinct layers were obtained from the 6 odourless liquid filled soft gel capsules. The ethanolic layer was carefully removed for testing.
P-glycoprotein Assay Procedures
Recombinant human P-gp ( MDR1 ) membranes were obtained from GENTEST Corp. Natural product extracts were examined for their ability to effect ATPase activity by the method stated by GENTEST using a THERMOmax Microplate Reader set to 650 nm to measure the orthovanadate-sensitive release of phosphate. Verapamil (Sigma) was used as a positive inhibitory control.
Fluorometric Microtiter Plate Assay Procedures
Extracts were screened for their ability to inhibit CYP 2C9, 2C19, 2D6, and 3A marker substrates using an in vitro fluorometric microtiter plate assay (4) adapted and modified from the one reported by Crespi et al. (25) and GENTEST (26). Comparative testing of the CYP 2C9*1 and 2C9*2, 3A4, 3A5 and 3A7 were done with balanced amounts of specific activity and protein content using Gentest insect control. Briefly, assays were performed with 5 ml of extract in clear-bottom, opaque-welled microtiter plates (96 well, Corning Costar, model # CSOO-3632, Corning, NY). Control wells contained distilled water or 55% ethanol and NADPH (ß-nicotinamide adenine dinucleotide phosphate, reduced form; Sigma Chemical Co., St. Louis, MO) solution; blank wells contained distilled water or 55% ethanol and buffer solution (1:4.7 v/v mixture of 0.5 M potassium phosphate in DW, pH 7.4); test wells consisted of extract or pure compound and NADPH solution. Test-blank wells consisted of the corresponding extract, pure compound and buffer solution. Enzyme solution was added to all wells. All microsomes were stored at -80° C until used and were not subjected to more than 2 freeze-thaw cycles.
The microwell plates were incubated for 1 h at 37 ° C. All assays were performed under gold fluorescent lighting (Industrial Lighting, Ottawa, ON). Only data sets yielding the highest readings without saturation were used to calculate percent inhibition values.
All samples were prepared in triplicate with the resultant percent inhibition calculations based on the mathematical combinations for the differences in fluorescence between the test/test-blank wells and the mean difference between each control and blank well. Thus, 9 experimental values were achieved for each sample. Controls were run with every assay. Each assay was repeated at least once.
Data was analysed by a one-way ANOVA for each isozyme, followed by a pairwise comparison utilizing Dunnett's test or Tukey's multiple range test.
Results
Two different extraction protocols were required to screen the 10 lots of commercially available garlic products for their ability to affect the activity of CYP 2C9, 2C19, 2D6 and 3A4 mediated metabolism (Table 2).
The odourless garlic samples and oils were extracted using 3 volumes 55% ethanol/liquid capsular material. The representative freeze dried garlic and aged garlic extracts were prepared as 25 mg/ml stock solutions. With only one exception, the odourless products had similar inhibitory profiles with 2C9 (21-33%), 2C19 (21-37%) and 3A4 (31-56%) (Table 2). The aged garlic extract and oils exhibited little influence on 2C19 activity. The freeze-dried tablet formulation was only inhibitory towards 3A4 (95.8%).
Table 2. The effect of commercial garlic products on cytochrome P450 isoform-mediated metabolism of marker substrates (n > 6; mean percent inhibition ± SD)
Product No
2C9
2C19
2D6
3A4
Odourless
1
33.5 ± 3.64
30.0 ± 5.26
4.8 ± 2.01
53.8 ± 2.15
2
21.4 ± 5.24
28.5 ± 4.95
1.8 ± 2.71
39.1 ± 3.31
3
23.4 ± 2.60
36.6 ± 6.19
5.0 ± 0.63
30.8 ± 4.42
4
32.4 ± 3.50
25.6 ± 3.05
7.5 ± 2.21
42.5 ± 3.84
5
31.5 ± 5.68
21.0 ± 11.47
6.9 ± 2.75
46.3 ± 4.19
6
4.8 ± 2.17
53.2 ± 5.25
4.4 ± 1.43
55.7 ± 5.85
Oil
7
22.2 ± 1.79
NA1
4.9 ± 1.05
31.3 ± 2.13
8
1.4 ± 2.43
NA
5.0 ± 0.62
17.4 ± 1.47
Tablet
9
NA
5.1 ± 4.13
6.5 ± 0.35
95.8 ± 0.81
Capsule
10
31.3 ± 2.83
NA
8.7 ± 0.93
44.2 ± 3.62
1NA, no inhibitory activity detected
To gain a better understanding of the chemical characteristics of the components in the aged garlic extract and the 3 fresh varieties, samples were extracted as above using solvents with increasing solvent strength values (Table 3.).
Table 3. Inhibition of cytochrome P450 using sequential solvent extracts (n > 6, mean percent inhibition ± SD)
Garlic
CYP
Hexane
Chloroform
Methylene Chloride
Ethyl Acetate
Methanol
Aged
3A4
37.2 ± 59.6
67.8 ± 31.8
67.9 ± 5.2
83.2 ± 2.8
54.0 ± 7.3
Chinese
3A4
2C1991.3 ± 9.8
48.5 ± 23.4116.1 ± 18.9
63.4 ± 18.090.7 ± 5.2
54.6 ± 13.085.1 ± 5.8
28.6 ± 16.648.2 ± 9.0
NA1Common
3A4
2C9135.8 ± 36.1
82.2 ± 12.692.9 ± 23.3
79.4 ± 10.565.0 ± 8.0
13.5 ± 23.672.9 ± 5.0
41.0 ± 23.658.6 ± 14.3
72.8 ± 2.4Elephant
3A4
2D687.0 ± 6.2
4.2 ± 0.9672.0 ± 24.8
12.6 ± 0.9793.4 ± 5.0
1.6 ± 4.293.3 ± 3.3
3.5 ± 0.9592.0 ± 5.6
4.8 ± 2.31NA, no inhibitory activity detected.
Extracts were reduced to dryness and reconstituted into methanol prior to testing for their effect on 3A4 mediated metabolism. The results show that all extract fractions exhibit a high inhibitory activity against the isoforms studied. As the results for the fresh garlic with 3A4 were similar, a representative sample was then screened for its effect on 2C9, 2C19 and 2D6 mediated metabolism (Table 3.). There was considerable varietal variation. Relatively strong inhibition occurred with both Chinese and common garlic on 2C19 and 2C9 mediated metabolism, respectively, for all extracts except the Chinese methanolic extract. As expected from the findings with the garlic products, all extracts from the elephant garlic exhibited a weak influence on 2D6 activity. Selective pH extraction of the Chinese bulb garlic showed significant (50-80%) inhibitory activity in the strong acid, weak acid, neutral and basic fractions (data not shown) against 3A4.
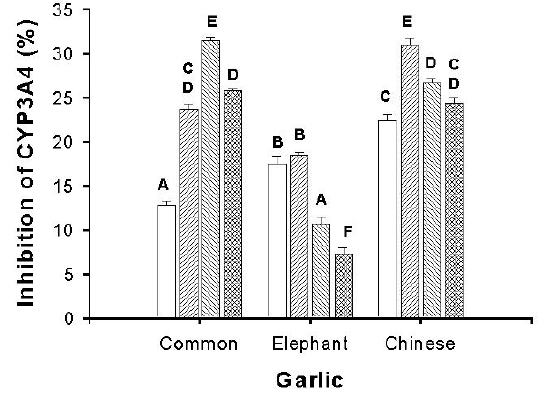
As differences were previously noted in the inhibitory effect of aqueous and methanolic extracts of fresh and aged garlic cloves on 3A4 mediated metabolism, the 3 varieties were extracted under 4 different conditions (Figure 1).
Figure 1: Effect of 3 different fresh garlic preparations made in various solvents (25 mg/ml) on the metabolism of the cytochrome P450 (CYP) 3A4 marker substrate 7-benzyloxyresorufin. Solvents are water (O), phosphate buffer (//), methanol (\\), and 55% ethanol (X). Data were analysed by a two-way ANOVA (n = 9; mean ± SEM) followed by Tukey=s multiple range test. Means with the same letter are not significantly different (p > 0.05).
Results varied with variety, but in general, the distilled water and phosphate buffer extracts gave the strongest overall suppression effect by the isoform-mediated metabolism of the marker substrates. Phosphate buffer extracts of the varietal frozen stock solutions were screened for their effect on metabolism mediated by the CYP isoforms 3A4, 3A5 and 3A7 (Table 4.).
Table 4. Inhibition of various cytochrome P450 3A isoforms by phosphate buffer extracts of fresh garlic ( n > 6; mean percent inhibition ± SD)
Garlic
3A4
3A5
3A7
Common
58.8 ± 2.2
42.9 ± 4.0
33.5 ± 5.9
Chinese
39.7 ± 1.1
19.4 ± 2.9
15.7 ± 2.8
Elephant
43.6 + 1.6
38.4 ± 4.5
10.3 ± 7.1
7BR was metabolized to a greater extent by 3A7 than by 3A4 or 3A5 with 3329, 1979 and 252 fluorescence counts, respectively. The 3A4 results for all extracts were consistent with earlier 3A4 findings (Table 3.). Extracts from common garlic exhibited a similar inhibitory effect on all 3A isoforms. Chinese and elephant garlic had a lesser inhibitory effect on 3A7; Chinese garlic extracts also had a lesser effect on the 3A5 isoform studied.
Figure 2: Activity of 3 different fresh garlic extracts on the metabolism of cytochrome P450 isozyme marker substrates: 7-methoxy-4-trifluoromethyl-coumarin (2C9*1, 2C9*2), 7-ethoxy-3-cyanocoumarin (2C19), 3-[2-(N,N-diethyl-N-methylamino)ethyl]-7-methoxy-4-methylcoumarin (2D6), and 7-benzyloxyresorufin (3A4). Bars correspond to control (O), common garlic (//), elephant garlic (\\), and Chinese garlic (X) fractions. Data was analysed by a one-way ANOVA (n $ 9; mean " SEM) for each isozyme, followed by a pairwise comparison utilizing Dunnett=s test (* indicates a significance of p < 0.050; ** indicates a significance of p < 0.001).
Extracts from the 3 fresh garlic varieties were then screened for their effect on CYP 2C9*1, 2C9*2, 2C19, 2D6 and 3A4 mediated-metabolism (Figure 2). The results are depicted as fluorescence counts to indicate formation of the marker substrate metabolite. 7-MFC was a better substrate for the 2C9*2 isoform. All 3 varieties had a slight inhibitory effect on 2C9*1-mediated metabolism, but highly stimulated metabolism of the marker substrate with the 2C9*2 isoform. The extracts had negligible to no effect on 2C19 and 2D6-mediated metabolism. However, all extracts strongly inhibited 3A4-mediated metabolism.
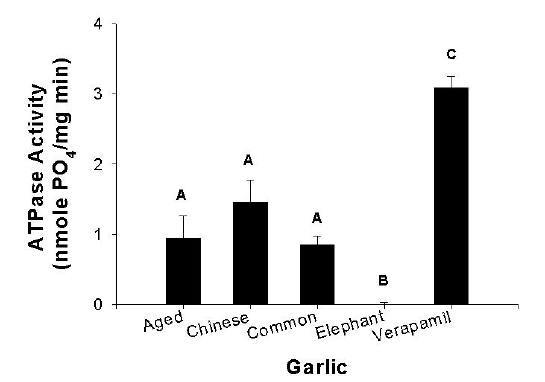
The effects of aqueous extracts from aged garlic capsules and the 3 fresh varieties were examined for their ability to interact with human P-gp membranes (Figure 3). Relative to 20 mM verapamil as the positive control, the aged, common, and Chinese phosphate buffer extracts had moderate levels of product-stimulated vanadate-sensitive ATPase activity. Elephant garlic was inactive.
Figure 3: P-glycoprotein-ATPase activity of aqueous extracts of aged garlic capsules and 3 fresh garlic varieties (25 mg/ml) compared with 20 μ M verapamil as the positive control. Data was analysed by a one-way ANOVA (n = 9; mean ± SEM) followed by Tukey's multiple range test. Means with the same letter are not significantly different (p > 0.05).
Discussion
The findings in this study with extracts of fresh garlic, different brands and lots of odourless garlic, together with representative samples of garlic oil, freeze dried garlic, and aged garlic have confirmed our earlier report on the inhibitory effect of common and odourless garlic on the in vitro 3A4-mediated metabolism of testosterone (16). Most of the extracts demonstrated an inhibitory effect on the metabolism of marker substrates by the other P450 isoforms examined, the stimulatory effects of the extracts of the fresh garlic on CYP2C9*2 mediated metabolism of the marker substrate are noteworthy.
Expression of the human MDR1 gene in the commercial membrane preparations generates a vanadate-sensitive ATPase that is markedly stimulated by drugs known to interact with P-gp (27). As a preliminary screening tool, the results from the assay demonstrated that aged and fresh varieties of garlic have stimulated the activity to a lesser extent than by the positive inhibitor verapamil. As stimulation of the ATPase assay is correlated with increasing P-gp inhibition, this suggests that constituents in the aged, common, and Chinese garlics may be a substrate for P-gp transport and thus potential inhibitors.
The results of various pH and solvent strength extracts emphasized that a single organic or aqueous extract may provide representative and qualitative data on the isoforms examined but will underestimate the total inhibitory potential of the product. CYP3A4 mediated-metabolism was markedly inhibited by the solvents extracts. In one replicate the control blank background values from a different bulb of garlic were markedly lower. As these values were not excluded, values of greater than 100% inhibition were obtained resulting in a wide deviation for the combined results for these samples. This emphasizes the variability and difficulties when working with natural products. Unlike conventional single active ingredient therapeutic products, natural products such as garlic are complex mixtures that have inherent variation from several sources including: climate (sunlight, rain, fog, cloud, frost, hail, etc.); growth conditions (species, region, soil conditions); harvest and storage conditions; manufacturing and compounding process; and the presence of non-active conjugates which need to be converted to the active moiety. In addition, there are individualistic variations in the amount taken, form (pills, tinctures, teas, etc.), manner of preparation, length of use (first time or repeated use), combination with other products, genetic capacity, and health status of the user.
The active substance or substances of garlic that inhibited the marker substrate metabolism in this study are not known. These findings are consistent with reports that in addition to sulfur-containing compounds, garlic also contains numerous flavinoids/isoflavinoids (such as nobiletin, quercetin, rutin, and tangeretin), polysaccharides, prostaglandins, saponins, and terpenes (such as citral, geraniol, linalool, alpha- and b-phellandrene) (28 and 29). Many of these compounds are also present in onions and shallots (15).
Constituents of garlic may not need to be present in high levels to elicit a pharmacological effect in order to produce a systemic or pre-systemic effect on drug disposition. The potential for the garlic products examined in this study to affect drug disposition may increase if used in combination with one or more conventional therapeutic products or other natural products. Some herbal products such as angelica, anise, arnica, asafoetida, bogbean, boldo, capsicum, celery, chamomile, clove, danshen, fenugreek, feverfew, ginger, ginkgo, ginseng, horse chestnut, horseradish, licorice, meadowsweet, onion, papain, passionflower, poplar, quassia, red clover, turmeric, and others can also contain substances which may enhance or inhibit the effect of garlic (30). Patients on a polypharmacy regimen might be expected to be at a higher risk of a clinically significant event.
Liu (31) discussed how many natural products exhibited a biphasic effect with an initial inhibitory effect or an inductive effect when given 24 hr prior to testing. Results from our in vitro study indicate that the disposition of drugs metabolized by CYP 3A, the 2C subfamily of enzymes, and transport by P-gp could be inhibited after the co-administration with garlic or garlic products. The trend towards reduced plasma exposure of ritonavir after 4 days of concurrent therapy with odourless garlic capsules suggests that garlic may have induced the metabolism of ritonavir (17), but the duration of garlic therapy in healthy subjects may have been too short to observe a significant inductive effect. In 2 heart transplant patients, 3 weeks of St. John's wort (300 mg capsules taken three times daily) reduced cyclosporine levels to below therapeutic concentrations, which resulted in acute transplant rejection (11). A subsequent clinical trial with a 14 day exposure to St. John's wort in healthy volunteers caused a marked (57%) reduction in indinavir plasma concentrations (10). Similarly, the inhibitory actions of tinctures and pure compounds from other natural products (4) may be offset by inductive effects during repeated usage. Together, these reports suggest that single and repeated use of natural products with drugs may affect the pharmacokinetics of co-administered drugs in healthy subjects versus patients differently, depending on the duration of administration of the natural product, the health status, and the genetic disposition of the individual.
Conclusion
These in vitro findings demonstrate that garlic components can affect cytochrome P-450 2C, 2D and 3A mediated-metabolism. The short- and long-term effects of fresh garlic and garlic products on enzyme induction or inhibition, and clinical outcome require further study.
Acknowledgements
This research was supported in part by the Canadian Foundation for HIV Research, the AIDS Program Committee of Ontario and the National Science and Engineering Research Council of Canada (Strategic Program).
References
Ness, J., Sherman, F. T., Pan, C. X.: Alternative medicine: what the data say about common herbal therapies. Geriatrics. 54(10): 33-8, 40, 43, 1999.
Barrett, B., Kiefer, D., Rabago, D.: Assessing the risks and benefits of herbal medicine: an overview of scientific evidence. Altern. Ther. Health Med. 5: 40-9, 1999.
Ameer, B., Weintraub, R. A.: Drug interactions with grapefruit juice. Clin Pharmacokinet. 33: 103-121, 1997.
Budzinski, J., Foster, B. C., Vandenhoek, S., and Arnason, J. An In Vitro Evaluation of Human Cytochrome P450 3A4 Inhibition by Selected Commercial Herbal Extracts and Tinctures. Phytomedicine. 7(4):273-282. 2000.
Eagling, V. A., Profit, L., Back, D. J.: Inhibition of the CYP3A4-mediated metabolism and P-glycoprotein-mediated transport of the HIV-1 protease inhibitor saquinavir by grapefruit juice components. Br J Clin Pharmacol. 48(4): 543-552, 1999.
Foster, B. C., Vandenhoek, S., Hanna, J., Akhtar, M. H., Krantis, A.: Effect of natural health products on cytochrome P-450 drug metabolism. Can J Infectious Dis Supp. 10 (B): 18, 1999.
Kivisto, K. T., Lilja, J. J., Backman, J. T., Neuvonen, P. J.: Repeated consumption of grapefruit juice considerably increases plasma concentrations of cisapride. Clin Pharmacol Ther. 66(5): 448-53, 1999.
Laroche M, Choudhri S, Gallicano K, Foster b. Severe gastrointestinal toxicity with concomitant ingestion of ritonavir and garlic. Can J Infectious Dis Supp. 9 (B): 471P, 1998.
Nebel, A., Schneider, B. J., Baker, R. K., Kroll, D. J.: Potential metabolic interaction between St. John's wort and theophylline. Ann Pharmacother. 33: 502, 1999.
Piscitelli, S. C., Burstein, A. H., Chaitt, D., Alfaro, R. M., Falloon, J.: Indinavir concentrations and St. John's Wort. The Lancet. 355: 547-548, 2000.
Ruschitzka, F., Meier, P. J., Turina, M., Luscher, T. F., Noll, G.: Acute heart transplant rejection due to St. John's Wort. The Lancet. 355: 548-549, 2000.
Taylor, J. R., Wilt, V. M.: Probable antagonism of warfarin by green tea. Ann Pharmacother. 33: 426, 1999.
Miller, L. G.: Herbal medicinals: selected clinical considerations focusing on known or potential drug-herb interactions. Arch Intern Med. 158(20): 2200-11, 1998.
Lawson, L. D., Wang, Z-Y. J., Hughes, B. G.: Identification and HPLC quantification of the sulfides and dialk(en)yl thiosulfinates in commercial garlic products. Planta Med. 57: 363-370, 1990.
Block, E., Naganathan, S., Putman, D., Zhao, S-H.: Allium chemistry: HPLC analysis of thiosulfinates from onion, garlic, wild garlic (Ransoms), leek, scallion, shallot, elephant (great-headed) garlic, chive, and Chinese chive. Uniquely high allyl to methyl ratios in some garlic samples. J. Agric. Food Chem. 40: 2418-2430, 1992.
Foster, B. C., Gallicano, K. D., Cameron, W., Choudri, S. H.: Constituents of garlic inhibit cytochrome P-450 3A4 mediated drug metabolism. Can J Infectious Dis Supp. 9 (B): 472, 1998.
Choudri, S. H., Gallicano, K., Foster, B., LeClaire T. A study of pharmacokinetics interactions between garlic supplements and ritonavir in healthy volunteers (abstract no. 1637). In: Program and Abstracts of the 40th Interscience Conference on Antimicrobial Agents and Chemotherapy, 2000 Toronto, Canada. p. 332.
Tsen, L. C., Segal, S., Pothier, M., Bader, A. M.: Alternative medicine use in presurgical patients. Anesthesiology. 93(1): 148-51, 2000.
Hadley, S. K., Petry, J. J.: Medicinal herbs: A primer for primary care. Hosp Pract. 34(6): 105-6, 109-12, 115-6, 1999.
Kwak, M. K., Kim, S. G., Kim, N. D.:Effects of garlic oil on rat hepatic P4502E1 expression. Xenobiotica. 25: 1021-29, 1995.
Siess, M. H., Le Bon, A. M., Canivenc-Lavier, M. C., Suschetet, M.: Modifications of hepatic drug-metabolizing enzymes in rats treated with alkyl sulfides. Cancer letters. 120: 195-201, 1997.
Brady, J. F., Ishizaki, H., Fukuto, J. M., Lin, M. C., Fadel, A., Gapac, J. M., Yang.: Inhibition of cytochrome P-450 2E1 by diallyl sulfide and its metabolites. Chem Res Toxicol. 4: 642-47, 1991.
Teyssier, C., Guenot, L., Suschetet, M., Siess, M. H.: Metabolism of diallyl disulfide by human liver microsomal cytochromes P-450 and flavin-containing monooxygenases. Drug Metab Disposit. 27: 835-41, 1999.
Oelkers, B., Diehl, H., Liebig, H.: In vitro inhibition of cytochrome P-450 reductases from pig liver microsomes by garlic extracts. Arzneimittelforschung. 42: 136-39, 1992.
Crespi, C. L., Miller, V. P., Penman, B. W.: Microtiter plate assays for inhibition of human, drug-metabolizing cytochromes P450. Anal Biochem. 248: 188-190, 1997.
GENTEST: Methodology of a high throughput method for measuring cytochrome P450 inhibition (version 2). Woburn: GENTEST Technical Bulletin , GENTEST Corporation, 1998.
Sarkadi, B., Price, E.M., Boucher, R.C., Germann, U.A., Scarborough, G.A.: Expression of the human multidrug resistance cDNA in insect cells generates a high activity drug-stimulated membrane ATPase. J Biol Chem. 267(7): 4854-4858, 1992.
Newall, C. A., Anderson, L. A., Phillipson, J. D. (eds.), Herbal Medicines: A guide for health-care professionals. The Pharmaceutical Press, London, pp 129-133, 1996.
Iberl, B., Winkler, G., Muller, B., Knobloch, K.: Quantitative determination of allicin and alliin from garlic by HPLC. Planta Medica. 56: 320-26, 1990.
Jellin, J.M., Batz, F., Hitchens, K.: Garlic. Pharmacist's Letter/Prescriber's Letter Natural Medicines Comprehensive Database, Therapeutic Research Faculty, Stockton, CA, USA, pp. 407-409, 1999.
Liu, G.: Effects of some compounds isolated from Chinese medicinal herbs on hepatic microsomal cytochrome P-450 and their potential biological consequences. Drug Metab Rev. 23(3&4): 439-465, 1991.
Corresponding Author: Office of Science, Therapeutic Products Directorate, Health Canada, Holland Cross 3102C3, 1600 Scott Street, Ottawa, Ontario, Canada K1A 1B6. brian_foster@hc-sc.gc.ca
Published by the Canadian Society for Pharmaceutical Sciences.
Copyright © 1998 by the Canadian Society for Pharmaceutical Sciences.
http://www.ualberta.ca/~csps