J Pharm Pharmaceut Sci (www.cspscanada.org) 9(2):222-230, 2006
Atorvastatin has distinct effects on endothelial markers in different mouse models of atherosclerosis
Petr Nachtigal, Gabriela Jamborova, Nada Pospisilova, Katerina Pospechova, Dagmar Solichova, Petr Zdansky, Vladimir Semecky
Department of Biological and Medical Sciences, Faculty of Pharmacy, Charles University, Hradec Department of Metabolic Care and Gerontology, Medical School and Teaching Hospital, Charles University, Hradec Kralove, Czech Republic
Received May 29, 2006; Revised June 21, 2006; Accepted July 24, 2006; Published, July 27, 2006.
Corresponding Author: Dr. Petr Nachtigal, Department of Biological and Medical Sciences, Faculty of Pharmacy, Email: nachti@faf.cuni.cz;
ABSTRACT: PURPOSE: Atherosclerosis is a progressive process that initially involves endothelial dysfunction. We investigated the effects of atorvastatin on both lipid parameters, and VCAM-1 and ICAM-1 expression in apoE-deficient or wild type C57BL/6J mice. METHODS:The C57BL/6J mice were fed with either chow or an atherogenic diet for 12 weeks. Male apoE-deficient mice were fed with the chow diet for 12 weeks. In 3 atorvastatin treated groups mice were fed the same diet as described above except atorvastatin was added to the diet at the dosage of 10 mg/kg per day for the last 8 weeks before euthanasia. RESULTS: Biochemical analysis showed that atorvastatin significantly decreased total cholesterol levels and VLDL in C57BL/6J mice fed with atherogenic diet but increased serum lipid levels in apoE-deficient mice. Stereological analysis of the immunohistochemical staining revealed that atorvastatin reduced endothelial expression of ICAM-1 and VCAM-1 only in C57BL/6J mice on chow diet. CONCLUSION: We have demonstrated that endothelial expression of both VCAM-1 and ICAM-1 does not correlate with cholesterol levels in these mice. Moreover, we showed that 8-week administration of atorvastatin decrease endothelial expression of VCAM-1 and ICAM-1 in C57BL/6J wild type mice beyond its lipid lowering effect but not in C57BL/6J wild type mice fed by atherogenic diet or in apoE-deficient mice.
Introduction
Atherosclerosis is a progressive process that initially involves endothelial dysfunction and accumulation and peroxidation of intimal lipids, followed by release of inflammatory cells and growth factors, resulting in vascular smooth muscle cell (VSMC) proliferation and collagen matrix production [24]. Endothelial dysfunction followed by monocyte rolling and adhesion to the vascular endothelial lining and subsequent diapedesis are not only the first steps, but also seem to be crucial events in the atherosclerotic process [18]. Members of the immunoglobulin superfamily of endothelial adhesion molecules, vascular cell adhesion molecule (VCAM-1) and intercellular cell adhesion molecule (ICAM-1), strongly participate on leukocyte adhesion to the endothelium. VCAM-1 is highly expressed on endothelia prone to develop atherosclerosis in such atherosclerotic models as apoE-deficient mice (apoE−/−), LDL receptor - deficient mice and rabbits fed with an atherogenic diet [12]. ICAM-1 is expressed strongly on the endothelium overlying atheromatous plaque in human coronary and carotid arteries [7], hypercholesterolemic rabbits [12], apoE−/− and LDLR−/− mice [19], although it is expressed in virtually all endothelial cells.
The advent of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase inhibitors, or statins, has revolutionized the treatment of hypercholesterolemia. Statins competitively inhibit HMG-CoA reductase, the enzyme that catalyzes the rate-limiting step in cholesterol biosynthesis. In addition a growing body of evidence suggests that statins exert beneficial vascular effects that are independent of their cholesterol-lowering potencies [5].
It has been demonstrated that mice are highly resistant to atherosclerosis. However, when mice were fed by very high cholesterol, high-fat diet that also contained cholic acid, hypercholesterolemia is induced and atherosclerotic lesions are formed in the mouse aorta [20]. Moreover, other mouse models of atherosclerosis have been generated by gene knockout technology. ApoE-deficient (apoE-/-) mice, have been shown to develop pronounced hypercholesterolemia and atherosclerotic lesions [23] on a diet with normal fat content and with no cholesterol supplementation [33]. It has been demonstrated that statins have diverse effects on atherogenesis in various mouse models of atherosclerosis [30]. It has been shown that statins lower lipid levels in LDLR-/- mice [3], but not in mice [22] and wild type (WT) C57BL/6J mice [11]. Moreover, some conflicting data about statins effects on atherogenic changes in the vessel wall of apoE mice exist [25; 30].
Since above mentioned studies were made on older mice with advanced atherosclerotic lesions we focused only on the changes in the expression of some markers of endothelial dysfunction. We hypothesized whether atorvastatin affects the endothelial expression of VCAM-1 and ICAM-1 in apoE-/- and wild type C57BL/6J mice with different levels of cholesterol. Therefore, we investigated the effects of atorvastatin treatment on both lipid parameters, and on endothelial expression of VCAM-1 and ICAM-1 by means of immunohistochemistry and stereology.
The major findings of the present study are that atorvastatin has different effects on cholesterol levels and VCAM-1 and ICAM-1 endothelial expression in wild type C57BL/6J mice fed chow or atherogenic diet and apoE-/- mice.
METHODS AND MATERIALS
The Ethical Committee of the Faculty of Pharmacy, Charles University, approved the protocols of the animal experiments. The protocol of experiments was pursued in accordance with the directive of the Ministry of Education of the Czech Republic (No. 311/1997).
Experimental Animals
Male C57BL/6J mice and male homozygous apoE-deficient mice on a C57BL/6J background weighing 15-20 grams were kindly provided by Prof. Poledne (IKEM, Prague, Czech Republic) and housed in the SEMED (Prague, Czech Republic).
Experimental design
All mice were weaned at 4 weeks of age and randomly subdivided into six groups. The C57BL/6J mice (n=8) were fed with the chow diet or an atherogenic diet containing 1.25% cholesterol, 15% fat, and 0.5% cholic acid for 12 weeks after the weaning. Male apoE-deficient mice were fed with the chow diet for the same 12 weeks after the weaning. In three atorvastatin treated groups mice were fed the same diet as described above except atorvastatin was added to the diet at the dosage of 10 mg/kg per day for the last 8 weeks before euthanasia. Each mouse in atorvastatin treated groups lived in a separate cage obtaining 6 g of food (in especially prepared pellets) daily with water ad libitum throughout the study. The food consumption was monitored every day. No differences in the food consumption were visible neither among animals of one experimental group nor between experimental groups. The dose of atorvastatin used in the present study was based on the doses used in previous study with mice [8; 28].
At the end of the treatment period, all animals were fasted overnight and sacrificed. Blood samples were collected via cardiac puncture at the time of death. The aortas, attached to the top half of the heart, were removed and then immersed in OCT (Optimal Cutting Temperature) embedding medium (Leica, Prague, Czech Republic), snap frozen in liquid nitrogen cooled 2-methylbutane and stored at -80°C.
Biochemistry
Serum lipoprotein fractions were prepared using NaCl density gradient ultracentrifugation (Beckman TL 100, Palo Alto, CA). The lipoprotein fractions were distinguished in the following density ranges: very low density lipoprotein (VLDL) < 1.006 g/ml; low density lipoprotein (LDL) < 1.063 g/ml; high density lipoprotein (HDL) > 1.063 g/ml. Total concentration and lipoprotein fraction concentration of cholesterol were assessed enzymatically by conventional diagnostic kits (Lachema, Brno, Czech Republic) and spectrophotometric analysis (cholesterol at 510 nm, triglycerides, at 540 nm wavelength), (ULTROSPECT III, Pharmacia LKB Biotechnology, Uppsala, Sweden).
Immunohistochemistry
Sequential tissue sectioning started in the mouse heart until the aortic root containing semilunar valves together with the aorta appeared. From this point on, serial cross-sections (7μm) were cut on a cryostat and placed on gelatin-coated slides. Sections were air-dried and then slides were fixed for 20 minutes in acetone at -20°C. Endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide in phosphate buffered saline (PBS) for 15 minutes. After blocking of nonspecific binding sites with 10% normal horse serum (Sigma-Aldrich Chemie, Steinheim, Germany) in PBS solution (pH 7.4) for 30 min, slides were incubated with primary antibodies for 1 hour at room temperature. After a PBS rinse, the slides were developed with biotinylated horse-anti goat IgG antibody or donkey anti-sheep IgG, in the presence of 200microg/mL normal mouse IgG. Antibody reactivity was detected using HRP (horse radish peroxidase)-conjugated biotin-avidin complexes (Vector Laboratories, USA) and developed with diaminobenzidine tetrahydrochloride as substrate. Specificity of the immunostaining was assessed by staining with nonimmune isotype-matched immunoglobulins.
Primary antibodies included the following: monoclonal antibody Rat Anti-Mouse CD31 (platelet endothelial cell adhesion molecule, PECAM-1) monoclonal antibody diluted 1:100, Rat Anti-Mouse CD106 (VCAM-1) diluted 1:100, and monoclonal antibody Hamster Anti-Mouse CD54 (ICAM-1) diluted 1:200. All antibodies were purchased from BD Pharmingen (California, USA).
Quantitative analysis of the immunohistochemistry
Stereological methods for the estimation of immunohistochemical staining of VCAM-1, ICAM-1, and PECAM-1 were used as previously described [16; 17]. In brief, the systematic uniform random sampling and the principle of the point-counting method were used for the estimation [32]. A total number of 50 consecutive serial cross sections were cut into 7μm thick slices, which gave us 0.350 mm lengths of the vessel called the reference volume. This reference volume comprises several sections of the vessel containing semilunar valves in aortic root, and several sections of aortic arch (ascending part of the aorta). A systematic uniform random sampling was used in the reference volume. The first section for each immunohistochemical staining was randomly positioned in the reference volume and then each tenth section was used, thus five sections for each staining were used for the stereological estimation. The point-counting method was used and more than 200 test points per vessel, hitting immunostaining, were counted for an appropriate estimation [9]. The estimated area is then:
![]()
where the parameter a characterizes the test grid and P is the number of test points hitting either the atherosclerotic lesion or positive immunostaining.
The area of PECAM-1 expression was considered as a total area of intact endothelium. Thus, the area of VCAM-1, and ICAM-1 expression indicate the percentage of activated endothelial cells calculated as
![]() ,
,
where area (x) is the area of VCAM-1 or ICAM-1, in the endothelium and area (PECAM) is the area of PECAM-1 expression in the endothelium.
Photo documentation and image digitizing from the microscope were performed with the Nikon Eclipse E200 microscope, with a digital firewire camera Pixelink PL-A642 (Vitana Corp. Ottawa, Canada) and with image analysis software LUCIA version 5.0 (Laboratory Imaging, Prague, Czech Republic). Stereological analysis was performed with a PointGrid module of the ELLIPSE software (ViDiTo, Kosice, Slovakia).
Statistical analysis
All values in the graphs are presented as a mean ± SEM of n=8 animals. Statistical significance in the differences between groups was assessed by unpaired t-test with the use of the SigmaStat software (version 3.0). P values of 0.05 or less were considered statistically significant.
RESULTS
Biochemical analysis
Biochemical analysis showed that administration of atorvastatin to C57BL/6J mice fed with cholesterol and cholic acid resulted in significant decrease of total cholesterol levels (5.41 ± 0.35 vs. 4.11 ± 0.24 mmol/l, P = 0.005) and VLDL cholesterol levels (3.45 ± 0.38 vs. 1.99 ± 0.21 mmol/l, P = 0.002) in comparison to non-treated mice. Atorvastatin did not affect total cholesterol levels in WT C57BL/6J mice on chow diet (3.18 ± 0.36 vs. 2.69 ± 0.21 mmol/l, P = 0.277). Moreover, no effect of atorvastatin treatment on cholesterol fractions was detected in these mice as well. By contrast, in apoE-deficient mice, atorvastatin treatment significantly increased levels of total serum cholesterol (11.21 ± 0.75 vs. 17.51 ± 1.66 mmol/l, P = 0.005),
VLDL cholesterol levels (8.49 ± 0.65 vs. 14.33 ± 0.89 mmol/l, P = <0.001), LDL cholesterol levels (2.46 ± 0.30 vs. 3.51 ± 0.30 mmol/l, P = 0.036), and so HDL cholesterol levels (0.23 ± 0.05 vs. 0.40 ± 0.04 mmol/l, P = 0.029) in comparison to the non-treated mice (Fig. 1).
Immunohistochemical staining of VCAM-1 and ICAM-1 in the endothelium
PECAM-1 expression was observed only in endothelial cells in all groups of mice and this antibody was used as standard for the detection of intact endothelium. The expression of VCAM-1 and ICAM-1 was observed in vessel endothelium in all groups of animals (data not shown). Moreover the ICAM-1 expression was stronger then VCAM-1 in each experimental group. The expression of both VCAM-1 and ICAM-1 decreased only in C57BL/6J on chow diet treated with atorvastatin when compared to non-treated mice (Fig. 2 A-D). Atorvastatin treatment did not affect the expression of neither VCAM-1 nor ICAM-1 in both atherogenic diet fed animals and apoE-deficient mice (data not shown).
Stereological analysis of VCAM-1 and ICAM-1 immunohistochemical staining.
Quantitative stereological analysis of ICAM-1 and VCAM-1 endothelial expression was related to the PECAM-1 staining in endothelium, thus the results indicate the percentage of activated endothelial cells. It was demonstrated that PECAM-1 is not regulated by hypercholesterolemia thus it is used as marker of all (activated or non-activated endothelial cells) [19]. Atorvastatin treatment did not affect the expression of ICAM-1 in neither, mice fed by atherogenic diet (68.8 ± 5.6 vs. 68.4 ± 7.9%, P = 0.970) nor in apoE-deficient mice in comparison to non-treated mice (36.7 ± 5.1 vs. 49.4 ± 8.1%, P = 0.328) (Fig. 3). However, significant diminution of ICAM-1 (58.0 ± 3.6 vs. 43.8 ± 3.4%, P = 0.018) staining was observed in atorvastatin treated C57BL/6J mice on chow diet in comparison to the non-treated mice (Fig. 3). The results from stereological analysis of VCAM-1 staining were similar to the ICAM-1 expression. Atorvastatin significantly decreased VCAM-1 expression in C57BL/6J mice on chow diet only (37.3 ± 5.8 vs. 20.9 ± 6.4 %, P = 0.005) (Fig. 4). In contrast VCAM-1 staining was not affected by the atorvastatin treatment in neither mice fed by atherogenic diet (38.7 ± 6.6 vs. 41.0 ± 4.3%, P = 0.788) nor apoE-deficient mice (39.4 ± 5.1vs. 41.4 ± 6.6 %, P=0.959) (Fig. 4).
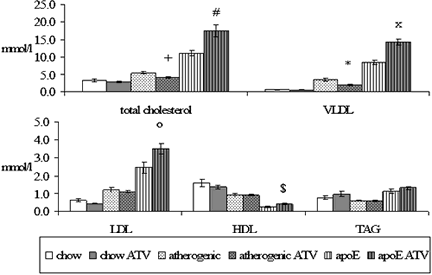
Figure 1: Serum lipid levels in all experimental mice. Atorvastatin significantly decreases total cholesterol levels (+P = 0.005 versus non-treated mice) and VLDL cholesterol (*P = 0.002 versus non-treated mice) in C57BL/6J mice fed with atherogenic diet. On the contrary, atorvastatin significantly increases levels of the total cholesterol (#P = 0.005 versus non-treated mice), levels of VLDL cholesterol (xP = <0.001 versus non-treated mice), levels of LDL cholesterol (oP = 0.036 versus non-treated mice) and HDL cholesterol ($P = 0.029 versus non-treated mice) in apoE-deficient mice.
. . . . . 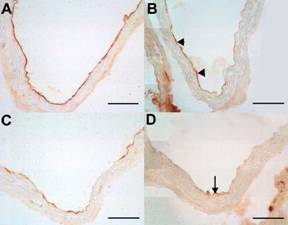
Figure 2: The immunohistochemical staining of endothelial expression of ICAM-1 (A, B), and VCAM-1 (C, D) in the aorta C57BL/6J mice on chow diet. Atorvastatin treatment notably reduced the expression of ICAM-1 (arrowhead) (B) when compared to non-treated C57BL/6J mice (A). VCAM-1 staining was markedly decreased (arrow) in atorvastatin treated animals (D). Bar = 50 μm.
. . . . . 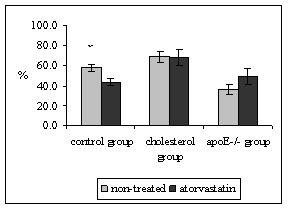
Figure 3: The percentage of activated endothelial cells for ICAM-1 in both aortic root and aortic arch. The expression of ICAM-1 was significantly decreased in C57BL/6J mice on chow diet after atorvastatin treatment (xP = 0.018 versus non-treated C57BL/6J mice). Atorvastatin treatment did not affect ICAM-1 staining in neither cholesterol fed animals, nor apoE-deficient mice.
. . . . . 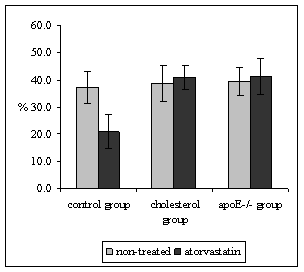
Figure 4: The percentage of activated endothelial cells for VCAM-1 in both aortic root and aortic arch. Atorvastatin treatment resulted in a significant diminution of endothelial expression of VCAM-1 in C57BL/6J mice on chow diet (xP = 0.005 versus non-treated C57BL/6J mice). VCAM-1 expression was not affected by the administration of atorvastatin in neither cholesterol fed mice, nor apoE-deficient mice.
DISCUSSION
A few years ago, several laboratories attempted to produce atherosclerosis in mice in order to identify potential modifier genes. Mice are highly resistant to atherosclerosis. On a low-cholesterol, low-fat diet, they typically have cholesterol levels of 2 mmol/l mostly contained in the antiatherogenic high density lipoprotein (HDL) fraction, and do not develop lesions. However, when mice were fed very high cholesterol, high-fat diet that also contained cholic acid, their cholesterol levels rose by a factor of two to three, with the majority now in the non-HDL fraction [4]. Moreover, genetic research and the application of transgenesis and gene targeting in mice resulted in generation of wide range of mice that are much more suitable atherosclerotic models including apoE-deficient mice, and LDLR-/- mice [10].
Clinical trials of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor therapy demonstrate an improvement in cardiovascular end points and coronary stenosis [25; 31].
However, it has been demonstrated the statins have various effects on cholesterol levels and atherogenic changes in the vessel wall in different mouse models of atherosclerosis [11; 30].
Thus, in this study we wanted to elucidate atorvastatin effects on both lipid parameters, and on endothelial expression of inflammatory markers VCAM-1 and ICAM-1 in WT C57BL/6J mice and apoE-deficient mice. The purpose was to reveal how atorvastatin affects the expression of VCAM-1 and ICAM-1 in these mice with different levels of cholesterol.
C57BL/6J mice are susceptible strain for the induction of atherosclerosis but diet saturated with fat, cholesterol and cholic acid must be used [21]. In our study, we found increased total serum cholesterol levels in group of mice after the feeding with diet containing 1.25% of cholesterol and 0.5% of cholic acid in comparision to mice fed by chow diet only. Several authors showed that prolonged feeding of this hyperlipidemic diet leads to the formation of atherosclerotic lesions which are restricted to the aortic root [20; 21]. However, we failed to detect any atherosclerotic lesions in neither cholesterol fed animals nor mice on chow diet in our study. This may be due to fact that period of feeding with atherogenic diet was not long enough in our study (12 weeks) in contrast to studies that used 14-18 weeks period of feeding [20].
ApoE- deficient mouse is a well-established genetic mouse model of atherogenic hypercholesterolemia, which is similar to hyperlipoproteinemia type III in humans. These mice lack their principal ligand for the LDL receptor and, therefore, develop hypercholesterolemia and atherosclerosis on diet with normal fat content [23]. We found the highest total serum cholesterol levels in apoE-deficient mice after 12 weeks on chow diet in comparison with C57BL/6J mice fed by either chow or atherogenic diet. Despite of this fact only small fatty streaks were visible in some mice in the aortic root.
Endothelial dysfunction followed by monocyte rolling and adhesion to the vascular endothelial lining and subsequent diapedesis are not only the first steps, but also seem to be crucial events in the atherosclerotic process [18]. Members of the immunoglobulin superfamily of endothelial adhesion molecules, vascular cell adhesion molecule (VCAM-1) and intercellular cell adhesion molecule (ICAM-1), strongly participate in leukocyte adhesion to the endothelium. The expression of VCAM-1 and ICAM-1 was largely studied by several authors in rabbit and mouse models of atherosclerosis [12; 13; 19]. It has been shown that VCAM-1 and ICAM-1 are detected in the regions predisposed to atherosclerotic lesion formation in normocholesterolemic rabbits, and the expression of both molecules is upregulated by a high-cholesterol diet in rabbits [12].Furthermore, Nakashima described that VCAM-1 is upregulated in apoE -/- mice on Western diet when compared to animals on chow diet [19]. On the contrary, in our study we found that endothelial expression of both ICAM-1 and VCAM-1 is almost the same in non-treated C57BL/6J on chow diet, atherogenic diet and apoE-/- mice in spite of the different cholesterol levels. These results are consistent with Zibara et al. who showed no significant differences in VCAM-1 and ICAM-1 endothelial expression between 16 weeeks old apoE-/- mice on chow diet and C57BL/6J on chow diet [34]. Thus we suggest that endothelial expression of both VCAM-1 and ICAM-1 is not so strongly correlated by cholesterol levels in mice as it was demonstrated in rabbits [13; 19].
Statins competitively inhibit HMG-CoA reductase, the enzyme that catalyzes the rate-limiting step in cholesterol biosynthesis. The resultant reduction in hepatocyte cholesterol concentration triggers an increased expression of hepatic LDL receptors, which clear LDL and LDL precursors from the circulation [27]. Moreover, recent experimental and clinical evidence indicate that some of the cholesterol-independent or “pleiotropic” effects of statins involve improving or restoring endothelial function, enhancing the stability of atherosclerotic plaques, decreasing oxidative stress and inflammation, and inhibiting the thrombogenic response in the vascular wall [1].
In our study, atorvastatin treatment did not affect total cholesterol levels in C57BL/6J mice on chow diet which is in acceptance of Choudhury et al. who showed that simvastatin has no effect on cholesterol levels in C57BL/6J mice on chow diet [11]. However, we found statistically significant diminution of endothelial expression of VCAM-1 and ICAM-1 in aortic root and aortic arch of these mice. Thus, we suggest that lipid independent effects of atorvastatin are responsible for the reduction of VCAM-1 and ICAM-1 expression. The same effects of statins on cell adhesion molecules expression were described in monkeys [26] and apoE*-Leiden mice [29].
However we must emphasize that despite this positive influence of atorvastatin on endothelium, these mice on chow diet do not develop atherosclerosis, thus they cannot be used as model for the studying of statins in early atherogenesis. On the contrary one may speculate about their usage as model for the study of pleiotropic effects of statins in endothelium in normocholesterolemic conditions.
The administration of atorvastatin significantly decreased total cholesterol levels in mice fed with diet containing 1.25% cholesterol, 15% fat, and 0.5% cholic acid. However, despite this hypolipidemic effect of atorvastatin we failed to detect any changes in endothelial expression of VCAM-1 and ICAM-1. The explanation of this fact could be associated with potential inclusion of cholate in the diet. It has been demonstrated that administration of cholate in the diet lead to the induction of oxidative stress with activation of NF-κB transcription factor. Thus, cholate in the diet can cause inflammation to the vascular endothelium and initiate the atherogenic cascade [14]. Moreover it has been demonstrated that multiple genes whose products are putatively involved in the atherosclerotic process are regulated by NF-κB. This includes E-selectin, monocyte chemoattractant protein-1 (MCP-1) as well as VCAM-1 and ICAM-1 [6]. Thus, we suggest that despite the significant hypolipidemic effect of atorvastatin in cholesterol fed mice, potential proinflammatory effects cholic acid on endothelium could mask the benefit of atorvastatin treatment. Therefore we propose that this animal model is not suitable for the studying of statins effects in endothelium.
Atorvastatin treatment (8 weeks) resulted in a paradoxical elevation in plasma total cholesterol in our study in apoE-deficient mice. Other authors showed the same increase of total cholesterol in apoE-deficient mice suggesting that statins might reduce lipoprotein clearance [2; 30]. These hypercholesterolemic effects of statins were accompanied by accelerated atherosclerosis [2] suggesting that anti-atherosclerotic effects of statins depend on the presence of apolipoprotein E [30]. In agreement with these studies we found that 8 weeks atorvastatin treatment had no effect on the expression of VCAM-1 and ICAM-1, thus no benefit of atorvastatin treatment was observed. On the contrary in our previous work we showed that 4 weeks administration of atorvastatin reduced endothelial expression of both VCAM-1 and ICAM-1 beyond its lipid lowering [15]. In addition, under similar experimental conditions, Sparrow et al. [25], showed that simvastatin significantly decreased aortic cholesterol accumulation without significantly decreasing plasma cholesterol levels. These experimental data suggest that experimental design of the study, especially the age of animals, and the duration of statins treatment, might be critical for their effect on the cholesterol levels and atherogenesis in apoE-deficient mouse.