J Pharm Pharmaceut Sci (www.cspscanada.org) 9(3):365-375, 2006
Atorvastatin attenuating down- regulation of peroxisome proliferator- activated receptor gamma in preventing cardiac hypertrophy of rats in vitro and in vivo
Ping. Ye1, Li. Sheng1, Cheng. Zhang1 and Yongxue. Liu2
1Department of Geriatric Cardiology, Chinese PLA General Hospital, Beijing China; 2Academy of Military Medical Sciences, PLA, Beijing, China.
Received September 11, 2006;Revised November 6, 2006; Accepted December 11, 2006; Published December 13, 2006
Corresponding Author. Department of Geriatric Cardiology, Chinese PLA General Hospital, Beijing 100853, China; E-mail: yeping301@yahoo.com.cn
ABSTRACT -- Purpose. To investigate whether the role of atorvastatin in suppression of cardiac hypertrophy is potentially associated with the change of peroxisome proliferator-activated receptor γ (PPARγ) expression, and the anti-inflammatory effect in vitro and in vivo. Method. Cardiac hypertrophy was established by angiotensin II in neonatal cardiac myocytes in vitro and incomplete ligation of abdominal aorta of SD rats in vivo. PPARγ and cytokines mRNA expression was evaluated by RT-PCR,and the rate of protein synthesis in cardiac myocytes by 3H-leucine incorporation. Results. Atorvastatin attenuated downregulation of PPARγ mRNA and inhibited brain natriuretic peptide (BNP), interleukin-1β (IL-1β) and matrix………….. metalloproteinase 9 (MMP9) mRNA expression, as well as 3H-leucine incorporation in a dose-dependent manner in vitro. Furthermore, atorvastatin reduced the mRNA expression of BNP, IL-1β and MMP9, and enhanced PPARγ mRNA expression,and diminished the pressure overload-induced increase in the ratio of heart weight to body weight, left ventricular wall thickness and myocyte diameter of rats in vivo. Conclusion. Atorvastatin prevents cardiac hypertrophy of rats, probably associated with the modulation of PPARγ and the inhibition of myocardial inflammation. Atorvastatin may play a role in prevention and treatment of cardiovascular diseases characterized by cardiac hypertrophy.
INTRODUCTION
Cardiac hypertrophy leading to heart failure is a major cause of morbidity and mortality worldwide. It represents a compensatory response to mechanical pressure overload and the change in neurohormoral factors and is observed in various cardiovascular diseases such as hypertension, myocardial infarction and valvular heart disease. Clinical studies have demonstrated that cardiac hypertrophy is not only an adaptation state before heart failure but also an independent risk factor for ischemia, arrhythmia and sudden death (1,2).
Studies performed in animal models of cardiac hypertrophy have shown that inflammatory cytokines, including interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α) and matrix metalloproteinase 9 (MMP9) are closely related to the occurrence and development of cardiac hypertrophy (3). As ligand-activated nuclear hormone receptor, peroxisome proliferator-activated receptors (PPARs) are recently found to play an important role in control of inflammatory response (4). PPARs consist of three isoforms, α, β/δ, and γ. PPARγ has been implicated in cardiac hypertrophy signaling (5). Recent studies indicate that activation of PPARγ negatively regulates inflammatory signaling pathway, (nuclear factor-κB and activator protein-1), subsequently attenuates angiotensin II- induced cardiomyocyte hypertrophy in vitro, as well as left ventricular hypertrophy in stroke-prone spontaneously hypertensive rats (6,7).
Statins or 3-hydroxy-methylglutaryl-CoA (HMG-CoA) reductase inhibitors are able to lower cholesterol by competitively inhibiting the activity of HMG-CoA reductase. They are widely prescribed as cholesterol-lowering agents that decrease the incidence of myocardial infarction and ischemic stroke. In addition, statins have some properties independent of its cholesterol-lowering effect, especially the anti-inflammatory effect. Recently statins have been shown to inhibit cardiac hypertrophy by cholesterol-independent mechanisms. Statins block the isoprenylation and activation of members of the Rho guanosine triphosphatase (GTPase) family, such as RhoA and Rac1, and the ability of statins to inhibit Rac1-mediated oxidative stress may make an important contribution to their inhibitory effects on cardiac hypertrophy (8). Moreover, Planavila et al (9) report that atorvastatin inhibits cardiac hypertrophy and prevents the fall in the protein levels of PPARα and PPAR β/δ and avoids NF-κB activation during cardiac hypertrophy. However, it is not well known whether the effect of statins on cardiac hypertrophy is related to PPARγ and to the inhibition on inflammatory cytokines. In the present study, thus, we examine whether the role of atorvastatin in suppression of cardiac hypertrophy is potentially mediated with the change of PPARγ expression, and the anti-inflammatory effect in vitro and in vivo.
MATERIALS AND METHODS
Materials
Dulbecco’s Modified Eagle’s medium (DMEM), calf serum and trypsin were purchased from Gibco (USA), angiotensin Ⅱ (AngⅡ) from Sigma (USA). RT-PCR kit, DNA Marker DL2000 were purchased from Takara (China), 3H-leucine from Beijing high tech Co., LTD (China), goat polyclonal antibody against PPARγ and FITC-labeled anti-goat antibody from Santa Cruz (USA). Atorvastatin was kindly provided by Pfizer Pharmaceuticals limited (China). Primers were synthesized by Beijing SBS Genetech Co., LTD (China).
Cell culture
Primary cultures of cardiac myocytes were prepared from SD rats (2~4 d) and performed according to method of Simpson (10). In the germfree condition, hearts were removed from SD rats and cut into pieces. The pieces were dispersed in 0.08% trypsin for approximately 5 min each. This digestion step was repeated several times until the pieces were digested completely and the collected cell suspensions were mixed with culture medium (DMEM with 10% calf serum). The dissociated cells were collected by centrifugation at 1000rpm for 8 min (4℃) and resuspended in DMEM supplemented with 10% calf serum. The resuspensions were collected in 50ml culture bottles and the nonmyocytes attached readily to the bottom of the culture bottle during the first hour. The resultant cell suspension was seeded in a new culture bottle/dish for 48 h at 37℃(5% CO2 in air ). During the first 48 h, 5-Bromo-2’-deoxyuridine (5-Brdu, 100μmol/L) was added to the culture medium to inhibit the proliferation of nonmyocytes. Then the culture medium was changed to serum-free DMEM and cells were pretreated with atorvastatin in different dosages (0.1, 1, 10μmol/L) for 30 min and subsequently stimulated with Ang II (1μmol/L) for 48 h.
Incorporation of 3H-leucine
Cultured neonatal ventricular myocytes, plated at 5x105 cells/ml in 12-well plates (1.5ml/well) were treated with atorvaststin and coincubated with 37kBq 3H-leucine for 48 h. The precipitated protein was collected on a fiberglass membrane. 3H-leucine incorporation was determined by LS-6500 liquid scintillometer for evaluating cardiomyocyte growth.
Pressure overload-induced cardiac hypertrophy
Thirty-two male Sprague-Dawley rats (180 to 200g) were maintained under standard illumination and temperature for 5 days before study began. The animals were randomly distributed into four groups as follows: control group (group A, n=8), sham-operated rats (group B, n=8), pressure overloaded rats without atorvastatin treatment (group C, n=8) and pressure overloaded rats treated with atorvastatin (group D, n=8). One week before the surgical procedure, the rats were administered with either atorvastatin (5mg/kg/day) or 0.9% salt solution by oral gavage and continued for 4 weeks after procedure. Pressure overload was induced by constriction of abdominal aorta by ligation with a blunted 25-gauge needle, which was then pulled out (6). For the age-matched sham-operation, the identical procedure was performed except that the suture was not tied around the aorta. The rats of all four groups were fed under the same condition. Hearts were harvested 4 weeks after operation. The heart weight/body weight (HW/BW) ratio was calculated and the heart samples were frozen in liquid nitrogen and then stored at -80oC. The study was approved by Standing Committee on Animal Research in our hospital.
Measurement of surface area of cardiac myocytes
The surface area of cardiac myocyte was measured according the method of Simpson (11). Cell images captured by video camera (Olympus) were traced and analyzed with NIH Image J software. One hundred cells were randomly selected from 2~3 flasks and surface area was measured.
Histological analysis
For histological analysis, all the hearts were arrested in diastole with KCl (30 mmol/L), followed by perfusion fixation with 10% formalin. Fixed hearts were embedded in paraffin, sectioned at 4-μm thickness, and stained with hematoxylin and eosin for overall morphology. The anterior, lateral, posterior walls as well as interventricular septum of the left ventricle 2 mm below the mitral valve were taken and the thickness was examined by Image-Pro Plus image analysis software. Then average thickness of the left ventricular walls was calculated. Mean myocyte diameter was calculated by measuring 100 cells from sections stained with hematoxylin and eosin.
RNA preparation and analysis
Total RNA was isolated by TRIzol reagent. RNA was quantified and the purity was assessed by absorbance at 260nm and 280nm respectively in DU-640 UV-spectrophotomer. The mRNA expression of brain natriuretic peptide (BNP), IL-1β, MMP9 and PPARγ from cultured ventricular myocytes and from left ventricle of rats were determined by RT-PCR. The primers used in RT-PCR were listed in table 1. The PCR was performed for 28~33 cycles. Each cycle consisted of denaturation at 95 oC for 45 s, primer annealing at 54~58oC for 1 min, and primer extension at 72oC for 1 min. The PCR product was separated using electrophoresis on 2% agarose gel and semi-quantified by an image analysis scanning system as a ratio to GAPDH.
Immunofluorescence staining
Myocytes plated on glass coverslips, were initially rinsed with DMEM medium for 30 s at ambient temperature and then fixed in 4% formaldehyde for 30 min. Cells were then washed with PBS for three times before permeabilization and after each subsequent step. Permeabilization was performed in buffer consisting of 1.0% Triton for 30 min at room temperature. Coverslips were sequentially incubated with goat polyclonal antibody against PPARγ and FITC-labeled anti-goat antibody, each for 60 min at room temperature. Cells were then visualized and photographed using a Zeiss fluorescence microscope (Bio-Rad, USA).
STATISTICAL ANALYSIS
Results are expressed as mean±SD. Difference among groups was tested by one-way ANOVA and that between two groups was tested by Dunnett T test. Both of one-way ANOVA and Dunnett T test were performed using SPSS statistical software.
RESULTS
Effect of atorvastatin on the surface area of cardiomyocytes
The surface area of AngII-stimulated myocytes increased significantly compared with normal myocytes and atorvastatin reversed the change obviously at a dosage of 10μmol/L (p<0.01), while DMSO, as a solvent control had no effect on the area of AngⅡ-stimulated myocytes. Atorvastatin had no effect on the area of normal myocytes (result was not shown) (Fig. 1).
Effect of Atorvastatin on 3H-leucine incorporation
Compared with normal myocytes, 3 H-leucine incorporation into AngII-stimulated myocytes increased significantly, and was inhibited by atorvastatin in a dosage-dependent way (p<0.01), but not by DMSO. However, the 3H-leucine incorporation was not altered by atorvastatin in normal myocytes (Fig. 2)
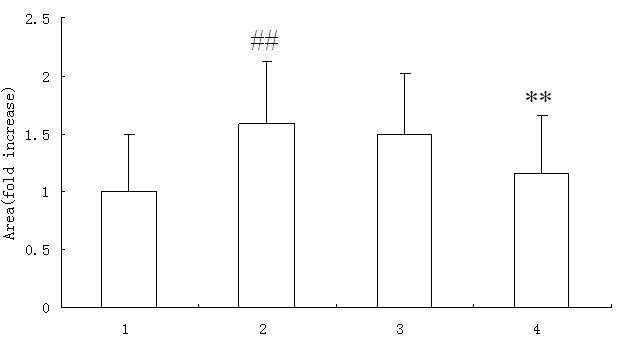
Figure 1. Effect of atorvastatin on the surface area of cardiac myocytes in vitro (n=100) Representative cardiac myocytes (X400). Cell images were captured by video camera and cell surface area was analyzed with NIH Image J software. 1. control; 2. AngII (1μmol/L); 3. AngII (1μmol/L)+DMSO; 4. AngII (1μmol/L) + atorvastatin (10μmol/L). Compared with control group # # p<0.01, compared with AngII group ** p<0.01
Effect of atorvastatin on pressure overload-induced left ventricular hypertrophy
Four weeks after operation of abdominal aortic coarctation (group C), the HW/BW ratio, average wall thickness of left ventricle, average diameter of cardiomyocyte all increased significantly when compared to sham group (group B) and control group (group A) (p<0.05). However, atorvastatin treatment markedly ameliorated the abnormalities in above parameters of group D in comparison to those in group C (Table 2) (p<0.05).
Effect of atorvastatin on PPARγ mRNA levels
The PPARγ mRNA expression was significantly downregulated when cardiac myocyte was stimulated with AngⅡ (p<0.01) in vitro. Atorvastatin markedly reversed the reduced PPARγ mRNA levels in a dosage-dependent manner (p<0.01), whereas DMSO had no impaction on PPARγ mRNA expression in AngⅡ-stimulated myocytes (Fig. 3). In left ventricle of pressure-overload rats in vivo, PPARγ mRNA expression was significantly suppressed as well compared with that in sham-operated rats and control rats (p<0.05), and was up-regulated by the treatment of atorvastatin (p<0.05) (Fig. 4).
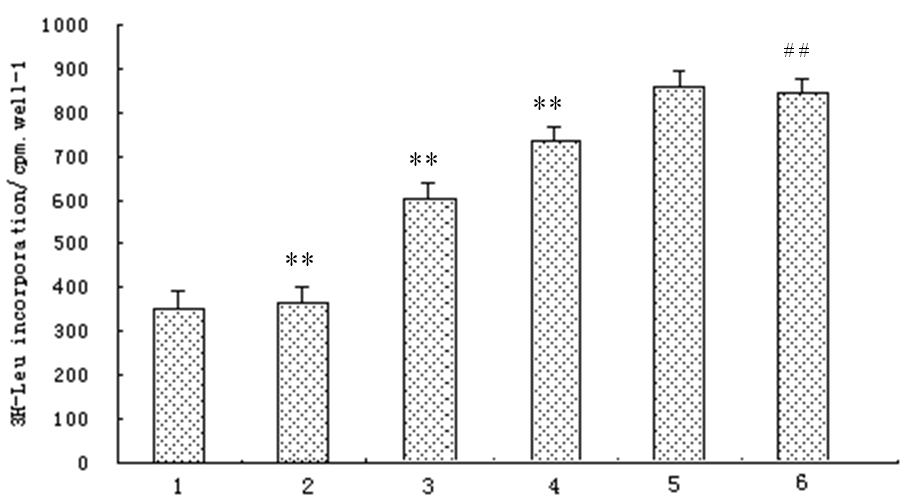
Figure. 2. Effect of atorvastatin on 3H-leucine incorporation in cardiac myocytes in vitro (n=3) 1.control; 2. AngII (1μmol/L)+Atorvastatin(10μmol/L); 3. AngII (1μmol/L)+Atorvastatin (1μmol/L); 4. AngII (1μmol/L)+Atorvastatin (0.1μmol/L); 5. AngII (1μmol/L)+DMSO; 6. AngII (1μmol/L); Compared with control group # # p<0.01, compared with AngII group **p<0.01.
Table 1. Oligonucleotide used in PCR amplification
genes |
Oligonucleotide sequence |
Fragment length(bp) |
BNP sense |
5’-TTTTCCTTAATCTGTCGCCG-3’ |
387 178 300 318 196 |
BNP: brain natriuretic peptide; GAPDH: glyceraldehyde-3-phosphate dehydrogenase;IL-1β: interleukin-1β; MMP9: Matrix metalloproteinases; PPARγ: peroxisome proliferator-activated receptorγ
Effect of atorvastatin on PPARγ protein levels in vitro
Immunofluorescence staining was applied to evaluate PPARγ protein level in AngII-induced cardiomyocytes with or without atorvastatin treatment. PPARγ was detectable in cytoplasm of controls, but markedly decreased after stimulation with AngII. PPARγ expression was improved (Fig. 5) and the protein was translocated from cytoplasm to nuclei (Fig.5 D) by treatment of atorvastatin.
Effect of atorvastatin on the expression of cytokines
In vitro study, the expression of BNP, IL-1β and MMP9 at mRNA level was markedly enhanced in AngⅡ-stimulated myocytes compared with normal myocytes (p<0.01) and significantly diminished by treatment of atorvastatin in a dose-dependent manner (p<0.01). DMSO had no effect on the mRNA expression of these genes (Fig. 6).
In vivo study, mRNA content of BNP, IL-1β and MMP9 in the left ventricle was significantly up-regulated in rats with left ventricular hypertrophy compared with sham-operated rats and control rats, and down-regulated obviously in rats treated with atorvastatin (p<0.05) (Fig. 4).
DISCUSSION
In the present study, we demonstrate that atorvastatin inhibits AngⅡ-induced neonatal rat cardiomyocyte hypertrophy in vitro and attenuates the degree of left ventricular hypertrophy induced by pressure overloaded in rats in vivo as well. These findings suggest that atorvastatin plays a potential role in the treatment of cardiovascular diseases characterized by left ventricular hypertrophy.
Inflammatory cytokine is closely related to the occurrence and development of cardiac hypertrophy. Dahl salt-sensitive rats develop hypertension with a high-salt diet and show left ventricular hypertrophy (LVH). The mRNA and protein content of inflammatory cytokines such as IL-1β, monocyte chemoattractant protein-1 (MCP-1) increase when LVH develops, and even further increase at the chronic heart failure (CHF) stage compared with that at LVH stage. The amount of IL-1β in the left ventricle is positively correlated with the left ventricular weight/body weight ratio (12). IL-1β may exert an impact on ventricular remodeling during transition from LVH to CHF. In vitro, IL-1β induces unique cardiomyocyte hypertrophy and marked secretion of atrial natriuretic peptide (ANP) and BNP, the marker gene for cardiac hypertrophy (13). Matrix metalloproteinases (MMPs) are members of a large family of zinc-dependent enzymes that degrade extracellular matrix. During the course of left ventricular remodeling, the synthesis and secretion of MMPs in mature left ventricular myocytes accelerate (14). The concentration of inflammatory cytokines in the heart changes markedly by the way of autocrine, which determines the direct role of cytokines in the occurrence and development of cardiac hypertrophy.
In the present study, it is found that the expression of BNP, IL-1β and MMP9 increases significantly in the rats when cardiac hypertrophy is induced both in vitro and in vivo. After treated with atorvastatin, the mRNA content of these inflammatory cytokines declines significantly. As expected, the declined cytokine level is associated with reduction in cardiomyocyte hypertrophy in vitro and left ventricular wall thickening in vivo. Atorvastatin is endowed with cardiac anti-hypertrophic properties and appears to be related to its capacity to decrease cardiac inflammatory cytokine concentration at transcription level.
Recently, the effect of statins on inflammatory cytokines and cardiac hypertrophy, as well as its therapeutic implication have been investigated. Ogata et al show that fluvastatin supresses IL-1β-induced cardiac hypertrophy in vitro (15). Nuclear factor-κB (NF-κB)、signal transducers and activation of transcription (STAT) and activator protein-1 (AP-1) are important transcription factors in inflammatory regulation.
Table 2. Effect of atorvastatin on left ventricular hypertrophy of rats
|
Group A |
Group B |
Group C |
Group D |
HW/BW(mg/g) |
3.62 ± 0.33 |
3.75 ± 0.31 |
4.77 ± 0.25 * |
4.20 ± 0.11*#a b |
Average wall thickness of left ventricle(mm) |
2.75 ± 0.41 |
2.98 ± 0.31 |
3.76 ± 0.37 * |
3.21 ± 0.35*# |
Average diameter of cardiomyocytes (μm) |
9.11 ± 1.35 |
9.58 ± 0.89 |
11.61 ± 1.34* |
10.31 ± 1.71*# |
Group A: control group (n=8), group B: sham-operated rats (n=8), group C: pressure overloaded rats without atorvastatin treatment (n=8) and group D: pressure overloaded rats treated with atorvastatin (n=8). HW/BW: heart weight/body weight. Compared with group D * p<0.05, compared with group B and group A # p<0.05
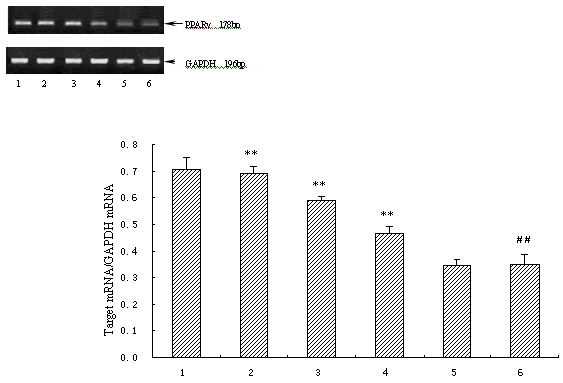
Figure 3. Effect of atorvastatin on PPARγ mRNA levels in cardiac myocytes in vitro (n=3) 1. control;2. AngII (1μmol/L)+atorvastatin (10μmol/L);3. AngII (1μmol/L)+atorvastatin (1μmol/L);4. AngII (1μmol/L)+ atorvastatin (0.1μmol/L);5. AngII (1μmol/L)+DMSO;6. AngII (1μmol/L). Compared with control group # # p<0.01, compared with AngII group ** p<0.01.
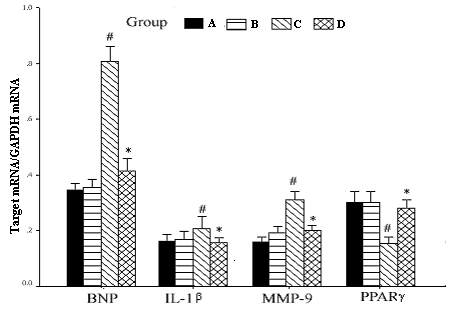
Figure 4. Effect of atorvastatin on the expression of inflammatory cytokines in vivo Group A: control group (n=8), group B: sham-operated rats (n=8), group C: pressure overloaded rats without atorvastatin treatment (n=8) and group D: pressure overloaded rats treated with atorvastatin (n=8). Compared with group C * p<0.05, compared with group A and B # p<0.05
...........................1.............. 2............... 3............... 4
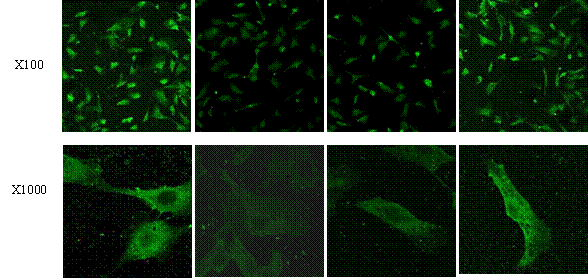
Figure 5. Effect of atorvastatin on PPARγ protein levels in vitro PPAR γ protein level was determined by immunofluorescence staining using a Zeiss fluorescence microscope. Representative cardiac myocytes were amplified by 100 times and 1000 times under fluorescence microscope respectively. 1: control; 2: AngII; 3: AngII+DMSO; 4: AngII+atorvastatin (10umol/L).
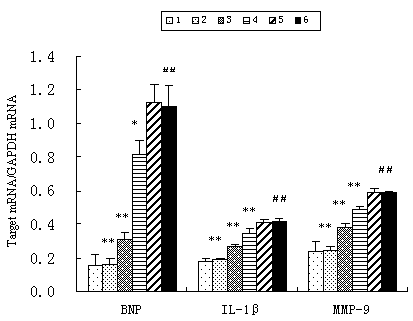
Figure 6. Effect of atorvastatin on the expression of inflammatory cytokines in vitro (n=3) 1. control;2. AngII (1μmol/L)+atorvastatin (10μmol/L);3. AngII (1μmol/L)+atorvastatin (1μmol/L);4. AngII (1μmol/L)+ atorvastatin (0.1μmol/L);5. AngII (1μmol/L)+DMSO;6. AngII (1μmol/L); Compared with control group # # p<0.01, compared with AngII group * p<0.05 ** p<0.01.
They are implicated in the regulation of inflammatory cytokine expression and exert therapeutic effect on left ventricular remodeling after myocardial infarction (16). Dechend et al demonstrate that statin inhibits the expression of inflammatory factors by suppressing transactivation of such nuclear factors as NF-κB and AP-1 in preventing cardiac hypertrophy of hypertensive rats (17).
PPARs belong to the nuclear receptor superfamily which are ligand-activated transcription factors. To date, three different PPAR subtypes have been identified: PPARα, PPARβ/δ and PPARγ (18). All these three PPARs are expressed in cardiovascular tissues, where they have several metabolic effects and increasingly recognized cardiovascular effect (19,20). It has recently been demonstrated that PPARγ negatively regulates the transcription of inflammatory response genes by antagonizing the AP-1, NF-κB and STAT signaling pathway (21). Moreover, Duan et al (22) report that cardiomyocyte-specific PPARγ knock-out mouse develops cardiac hypertrophy, although PPARγ expression is relatively lower compared with PPARα or PPARβ/δ. Also, treatment of Ang II-stimulated cultured cardiac myocytes and pressure overload-induced cardiac hypertrophy with PPARγ ligands--thiazolidinediones attenuates the cardiac hypertrophy in vitro and in vivo (6), consistent with our proposal that PPARγ inhibits cardiac hypertrophy.
It has also been found that PPARγ mRNA concentration is increased and IL-6 secretion is suppressed by atorvastain treatment in adipocytes of rabbits on high-cholesterol diet, indicating a possible negative relationship between PPARγ and IL-6 expression by atorvastatin (23). In addition, it is reported that pravastatin increases PPARγ levels, suppresses NF-κB transactivation and the generation of inflammatory cytokines in oxLDL-stimulated monocytes, suggesting the involvement of transcription factors such as PPARγ and NF-κB in the modulation of the inflammatory processes by statins (24). However, similar studies conducted in the heart are still in paucity. In the present study, atorvastatin concentration dependently increases PPARγ mRNA level, diminishes inflammatory cytokine levels and cardiac hypertrophy in vitro, as well as in vivo. It is presumed that the effect of atorvastatin on cardiac hypertrophy may partly through the mechanism of activating PPARγ, in relation to control of inflammatory response. However, the issue needs to be confirmed and further investigated.
Although statins are widely used to treat dyslipidemia for prevention and treatment of atherosclerosis, knowledge about the effect of statins on left ventricular hypertrophy and heart failure in humans is quite limited. A small scale clinical study demonstrates that the serum concentration of IL-6, TNF-α, as well as BNP decreases in dilated cardiomyopathy patients receiving simvastatin, accompanied with improved left ventricular ejection fraction and a lower New York Heart Association functional class (25). A larger observational study shows that statin therapy is associated with a lower risk of death in patients with severe heart failure, independent of serum cholesterol levels (26). Further investigation of potential benefits of statins in patients with ventricular hypertrophy and heart failure appears warranted.
In conclusion, the present study demonstrates that atorvastatin prevents down-regulation of PPARγ and inhibits cardiac inflammation during the course of suppressing cardiac hypertrophy of rats in vitro and in vivo.
ACKNOWLEDGMENTS
This study was supported by the Nature Science Foundation of China (No. 30270551 ).