Andrew Simmonds

Andrew Simmonds
Ph.D., University of Alberta
Professor and Chair
Office: 5-19A Medical Sciences BuildingLaboratory: 5-19 Medical Sciences Building
andrew.simmonds@ualberta.ca
Research Interests
Regulation of cell division and differentiation by Vestigial-like proteins
Our research group uses a combination of live-cell imaging, Drosophila genetics, cell-culture and molecular biology to study the regulation of critical cellular processes like peroxisome function, mitosis and cell differentiation.
Understanding the cellular mechanisms underlying peroxisome disorders using Drosophila.
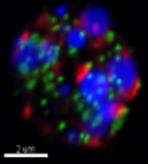 |
We use the fruit fly, as a simple animal model to study the basic processes that also occur in human cells and organs. For example, we previously used flies to discover new ways that peroxisomes function in human immune cells responding to microbial infection. We are currently characterizing cellular factors that coordinate lipid droplet storage and release of fat molecules and direct these fat molecules to peroxisomes. We will study isolated fly cells as well as cells within the organs of developing flies.
Regulation of cytoplasmic messenger RNAs in the developing embryo.
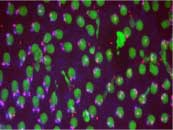 |
In eukaryotic cells, cytoplasmic processing (P-) bodies regulate directly regulate mRNAs. In mammalian cells, a core component of P-bodies is the GW182 family of proteins. The GW182 protein family has been linked to microRNA and small interfering RNA gene regulation. Our group was the first to identify and functionally confirm that there is only a single GW182-like protein (we have named Gw) in the genetic model system Drosophila melanogaster. Using a combination of cultured cells as well as studies in whole animals, we are attempting to dissect the cellular roles and cytoplasmic events that regulate mRNAs including the role of Gw-containing P-bodies. There is a long history of using Drosophila to establish the basic pathways of gene regulation, and in most cases, this information can be translated directly to mammalian cells (humans). The advantages of using a model system have already helped us to determine that loss of Gw function leads to uncoordinated nuclear (chromosome) divisions. Using the wealth of genetic and biochemical tools that have been developed specifically for working with Drosophila, we will address three main questions: 1) What is the role of cytoplasmic Gw during the post-transcriptional regulation of messenger RNAs? 2) Why is a mainly cytoplasmic protein like Gw required for successful chromosome division? 3) How is the production of Gw protein and formation of cytoplasmic GW-containing P-bodies regulated? With increased understanding of how this protein functions within the cell and how it, in turn, is regulated, our research will help with diagnosis and treatment of several poorly understood diseases that have been linked to GW182 in humans.
Regulation of myocyte division and differentiation by Vestigial-like proteins
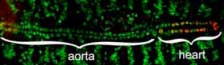 |
Our lab studies the Drosophila Vesigial proteins, a member of the conserved Vestigail-like family found in most higher metazoans. The Vestigal-like proteins interact with two DNA binding proteins (Scalloped and Myocyte Enhancing Factor-2) that have critical roles in establishing muscle cell fate. Vestigial-like proteins come in two types, oncogenic and tumour-suppressive. Both bind to Scalloped, which is directly regulated by the Hippo cell proliferation/differentiation pathway. There are multiple members of the human Scalloped and Myocyte Enhancing Factor-2 protein families and it has been extremely difficult to identify co-factors that regulate their activity. However in Drosophila melanogaster, there is only a single Scalloped and Myocyte Enhancing Factor-2
Recent Research
(The names of Simmonds lab members are indicated in bold)
Di Cara F, Sheshachalam A, Braverman NE, Rachubinski RA, Simmonds AJ. Peroxisome-Mediated Metabolism Is Required for Immune Response to Microbial Infection. Immunity. 2017 Jul 18;47(1):93-106
Pimmett VL, Deng H, Haskins JA, Mercier RJ, LaPointe P, Simmonds AJ. The activity of the Drosophila Vestigial protein is modified by Scalloped-dependent phosphorylation. Dev Biol. 2017 May 1;425(1):58-69.
Baron MN, Klinger CM, Rachubinski RA, Simmonds AJ. A Systematic Cell-Based Analysis of Localization of Predicted Drosophila Peroxisomal Proteins. Traffic. 2016 May;17(5):536-53.
Li J, Hobman TC, Simmonds AJ. Gawky (GW) is the Drosophila melanogaster GW182 homologue. Adv Exp Med Biol. 2013;768:127-45.
Mast FD, Li J, Virk MK, Hughes SC, Simmonds AJ, Rachubinski RA. A Drosophila model for the Zellweger spectrum of peroxisome biogenesis disorders. Dis Model Mech. 2011 Sep;4(5):659-72.
Schneider MD, Bains AK, Rajendra TK, Dominski Z, Matera AG, Simmonds AJ. Functional characterization of the Drosophila MRP (mitochondrial RNA processing) RNA gene. RNA. 2010 Nov;16(11):2120-30.
Deng H, Bell JB, Simmonds AJ. Vestigial is required during late-stage muscle differentiation in Drosophila melanogaster embryos. Mol Biol Cell. 2010 Oct 1;21(19):3304-1
Laboratory Members
Graduate Student
Kazuki Ueda
Andy Cheng
Technician
Julie Haskins