Transgenic Core Facility
What We Do
The University of Alberta Transgenic Core Facility was established to help researchers with their transgenic mouse production and archiving needs. It is located in the Health Sciences Laboratory Animal Services (HSLAS) facility.
Research Resource Identifier - RRID:SCR_019175
Services
Note: All requested strains must be on an approved Animal Use Protocol prior to commencement of work at the Transgenic Core Facility.
Mouse Sperm Cryopreservation
Sperm cryopreservation is an easy, inexpensive way to preserve your mouse lines of choice. Cryopreservation not only allows the researcher to stall breeding of lines not currently being used thereby reducing expenditure and animal usage and but it also prevents against disease (eg. pathogen outbreak) and genetic drift and allows for easier distribution of strains to other facilities. We request a single, proven-breeder male mouse (to ensure fertility), between 2-6 months of age. This provides enough sample for 8-10 straws to be frozen. Sperm will be checked for motility prior to and after cryopreservation. The cryopreservation service will include an in vitro fertilization (to 2-cell stage development) quality control step. Please note there will be a delay in getting IVF results. If you require results immediately, please consult with Serene Wohlgemuth.
Mouse in vitro Fertilization
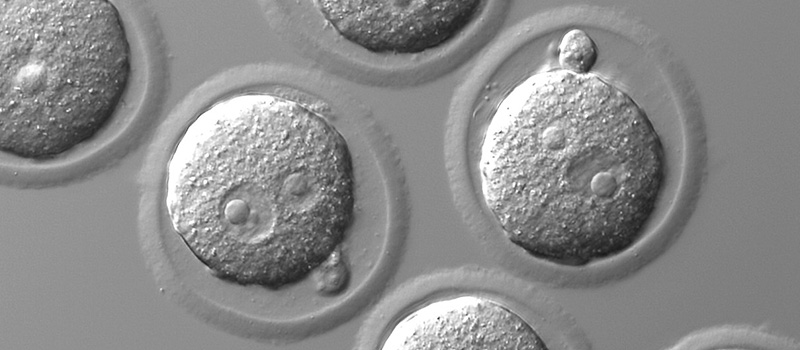
Mouse in vitro fertilization is usually used to recover strains that have been cryopreserved or to aid strains that have stopped breeding naturally. It is also used to rederive lines that have unknown health status or are known to have pathogens. Oocytes are collected from 3-5 superovulated females and incubated with sperm (either fresh or frozen) and cultured to the 2-cell stage after which the fertilized embryos are transferred to pseudopregnant CD1 females. The researcher will received all pups after weaning.
Pricing is dependent on the mouse strain required. A quote for animal costs will be provided by the core.
Mouse Rederivation
Rederivation of mice serves to remove pathogens from existing strains which may affect the researchers results and/or prevent them from maintaining the strain in the HSLAS facility, in the case of known excluded pathogens. To achieve this, an in vitro fertilization or breeding with superovulated female is performed using a male from the transgenic line of interest and wildtype commercially-available females. The resultant embryos are thoroughly washed to remove any potential contaminant and transferred to a pathogen-free female recipient. This female and any resultant pups will be housed separately to prevent cross-contamination into the HSLAS facility until health reports return confirming the animals are pathogen-free. The researcher will receive pups after this confirmation.
Pricing is dependent on the mouse strain required. A quote for animal costs will be provided by the core.
Transgenic Mouse Generation (DNA microinjection)
Transgenic mice are vital to many avenues of research, notably as a tool to model disease. The Transgenic Core Facility will inject your DNA construct into one of the pronuclei of newly fertilized oocytes and transfer the injected embryos into a pseudopregnant CD1 female. 3 round of this is performed (approximately 300 embryos injected). By default we use FVB animals but other strains may be used upon discussion. The researcher will received a tissue sample from each pups for genotyping and all identified founders will be transferred to the PI.
Pricing is dependent on the mouse strain required. A quote for animal costs will be provided by the core.
Equipment
- Hamilton Thorne Computer-Aided Sperm Analysis System
- Sutter Xenoworks Micromanipulator
- Sutter Xenoworks Digital Microinjector
- Nikon TE2000 Inverted Microscope
- Leica M165 Dissecting Microscope
- MINC Benchtop Incubator
- Sutter Micropipette Puller
- Fine Science Tools Bead Sterilizer
- Baker EdgeGUARD Clean Bench
- TMC Air Isolation Table
Transgenic Core User Committee
- Vacant - Academic Lead of the Transgenic Core Facility
- Dr. Matthew Macauley - Department of Chemistry, Faculty of Science
- Dr. Keith Fenrich - Faculty of Rehabilitation Medicine
- Dr. John Ussher - Faculty of Pharmacy & Pharmaceutical Sciences
- Dr. Serene Wohlgemuth - Manager, Transgenic Core Facility
- Dr. M. Joanne Lemieux - Vice-Dean, Research (Biomedical), FoMD
- Vacant - North Campus Animal Services Representative
- Colleen Sunderland - Manager, Core Research Facilities, FoMD